Topics:
States of matter:
States of matter, such as solid, liquid, and gaseous, are ways in which substances behave under different conditions. This applies not only in science, but also plays an important role in everyday applications, such as car air conditioning. In this story we take a closer look at what these states mean, especially in relation to car air conditioning. We use the refrigerant R134a as an example to make it clearer.
Substances can exist in different states: solid, liquid, and gaseous. This depends on how warm or cold it is and how much pressure there is. When the temperature is low, the substance is usually solid, like ice. At higher temperatures it becomes liquid, like water, and at even higher temperatures it changes into a gas, for example water vapor. This change of state is called phase transition.
In car air conditioning, understanding phase transitions is very important. The refrigerant, such as R134a, is crucial here. The refrigerant is constantly changing from liquid to gas and back again. While evaporating, the refrigerant absorbs heat and changes into gas. It is then compressed, causing it to return to liquid and release heat. This repeated cycle ensures that the temperature inside the car is properly regulated.
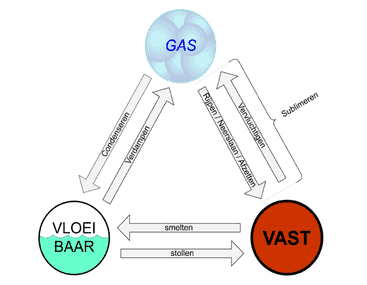
Condensation: Condensation is the process in which a gas changes to the liquid phase when it is cooled. In a car air conditioning system this is a crucial step to remove heat from the interior of the car and create a cooling effect. When the coolant, usually the refrigerant R134a or R1234YF, flows through the evaporator, it absorbs heat from the ambient air in the car. This heat absorption process cools the air in the interior. Because the coolant is exposed to relatively warm air at this point, it evaporates and changes into the gaseous phase. This gas is then drawn in by the compressor. In the compressor, the gaseous coolant is compressed, placing it under high pressure and significantly increasing its temperature. This hot, compressed gas phase of the coolant leaves the compressor and flows through the condenser.
Overheating: Overheating occurs when there is too little liquid coolant present in the system and the gas compressed by the compressor becomes warmer than normal. This can have several negative effects on the operation of the air conditioning system and the comfort of the passengers. If there is not enough liquid to remove the heat generated by the compressor, the gas that is forced through the compressor can reach an undesirably high temperature. This can lead to reduced cooling capacity and even damage to the compressor and other components in the system. Moreover, overheating can impair the efficiency of the entire air conditioning system. The system will have to consume more energy to reach the desired interior temperature, resulting in higher fuel consumption and possibly a reduced service life of the system.
Boiling temperature: This is equal to the condensation temperature and depends on the pressure. The boiling temperature is the temperature at which a vaporous substance begins to condense and revert to liquid form. In the case of an air conditioning system, the coolant, which is now in vapor form, is routed through the condenser section of the system. Here, heat is discharged to the environment, causing the coolant to cool and condense into a liquid. This condensation process ensures that the heat absorbed in the evaporator is now released to the environment outside the air conditioning system.
Saturated vapor: In an air conditioning system, there is saturated vapor when both the vapor and liquid of the same refrigerant are present. In other words, the saturated vapor is the vapor that is just beginning to condense at a certain temperature and pressure.
Subcooling: Subcooling occurs when the liquid in the coolant is colder than the boiling (condensation) temperature. This can cause various problems and negatively affect the performance of the air conditioning system. If the liquid is too cold, it can freeze when it flows through the evaporator. This can lead to ice formation on the evaporator and limited airflow, resulting in a loss of cooling capacity and even possible damage to the evaporator. Besides, subcooling can reduce the overall efficiency of the system. It can cause the system to take longer to reach the desired temperature, meaning passengers become comfortable less quickly in the vehicle.
