Introduction:
The name catalytic converter originally comes from the Greek word “katalysis” (which means decomposition). A catalytic converter has been required since the end of 1992 in order to comply with environmental regulations. Exhaust gases contain the harmful substances CO (carbon monoxide), NOx (nitrogen oxides) and CH (unburned hydrocarbons). These substances are (oxidized) into non-harmful substances. Hence the occasional term oxidation catalytic converter.
In chemistry, a catalyst is understood to be a substance that initiates and speeds up or slows down a chemical reaction without undergoing any change itself.

Operation of three-way / oxidation catalytic converter:
A catalytic converter is not a filter, but can be seen as a conversion element in which precious metals such as platinum, rhodium or palladium are applied. When the exhaust gases come into contact with them, a very rapid chemical reaction occurs. The molecules of the harmful gases are broken down and combined with other molecules, resulting in a non-harmful gas. The catalytic converter is capable of purifying the exhaust gases by 90%. This does, however, come at the expense of higher fuel consumption and lower power. That is because it creates a certain air resistance in the exhaust system.
Substances in the exhaust gases:
- CO2: Carbon dioxide (harmful to the environment, humans and animals in high concentrations)
- CO: Carbon monoxide (incompletely burned gas, also harmful to health)
- CH: Hydrocarbons (unburned petrol fractions)
- O2: Oxygen particles (that did not take part in the combustion)
- NOx: Nitrogen compound (that only occurs at very high combustion temperatures).
The catalytic converter converts the 3 harmful components CO, HC and NOx into 3 harmless components: CO2, H2O and N2. The name three-way catalytic converter also comes from this.
In order to add O2 and CO to the catalytic converter so that the conversion can take place, the injection pattern of the engine needs to be adjusted. To form O2, the mixture must be lean (less fuel, more air). To form CO, the mixture must be rich (more fuel, less air). The latter is not the case with lean-burn engines; see the chapter on the NOx catalytic converter further down the page.
By continuously injecting slightly too much and slightly too little fuel into the cylinders, a rich and lean mixture alternately occurs. The surpluses of CO and O2 thus end up in the catalytic converter. In the catalytic converter the platinum reacts with the CO and HC. The rhodium ensures the reduction of the NOx. That also explains why a varying voltage is measured when measuring on the lambda sensor. There the voltage varies between 0.2 and 0.8 volts (from lean to rich, etc.). The engine management system of the car (the ECU) controls this itself. So there is nothing to be adjusted.
| Harmful substance: | Adding: | Results in: |
| CO + | O2 = | CO2 |
| HC + | O2 = | CO2 + H2O |
| NOx + | CO = | N2 + CO2 |
In order to add O2 and CO to the catalytic converter so that the conversion can take place, the injection pattern of the engine needs to be adjusted. To form O2, the mixture must be lean (less fuel, more air). To form CO, the mixture must be rich (more fuel, less air). The latter is not the case with lean-burn engines; see the chapter on the NOx catalytic converter further down the page.
By continuously injecting slightly too much and slightly too little fuel into the cylinders, a rich and lean mixture alternately occurs. The surpluses of CO and O2 thus end up in the catalytic converter. In the catalytic converter the platinum reacts with the CO and HC. The rhodium ensures the reduction of the NOx. That also explains why a varying voltage is measured when measuring on the lambda sensor. There the voltage varies between 0.2 and 0.8 volts (from lean to rich, etc.). The engine management system of the car (the ECU) controls this itself. So there is nothing to be adjusted.
What can be seen in the table above is that the substances are all converted into, among other things, CO2. CO2 is nowadays considered to be a substance that is dangerous to the environment and responsible for global warming. However, a human also exhales CO2. This is converted back into O2 (oxygen) by trees and plants. An excess of CO2 has a harmful effect. Trees and plants are in the minority and are not able to convert everything into O2. For internal combustion engines, the CO2 content should be as high as possible. That sounds strange, because you would assume it should be kept as low as possible. The fact is this: the higher the CO2 content, the less CO and HC is released. CO and HC are directly harmful to health when inhaled. The only way to reduce the CO2 content is by switching to alternative fuels, smaller (more economical) internal combustion engines and a calmer driving style.
Operating temperatures:
The effective operation of the catalytic converter starts at a temperature of 250 degrees and is at its maximum at a temperature of 450 degrees. After starting the engine, it takes a while before the purifying effect begins. The catalytic converter is mounted as close as possible to the exhaust manifold, because that way it reaches its operating temperature sooner. Exhaust gas temperatures between 800 and 1000 degrees cause faster thermal aging, which shortens the service life and reduces the active surface area.
There are also catalytic converters with a heating element that ensures the catalytic converter reaches temperature even faster after a cold start. This allows it to start regulating even sooner after the engine has been switched on, resulting in cleaner exhaust gases.
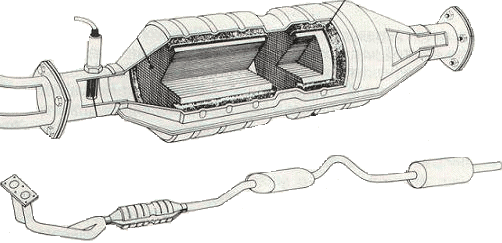
In order to warm up the catalytic converter as quickly as possible after a cold start, a secondary air pump is often used.
Operation of NOx catalytic converter:
It was explained earlier that NOx can be reduced by the catalytic converter by obtaining extra CO in the exhaust gas. This is only possible by enriching the mixture. In the lean-burn engines of, among others, Volkswagen (FSI) and BMW (Efficient Dynamics), the engines always run at part load and low engine speeds on a mixture with an excess of air (so lean, and never rich). With a normal three-way catalytic converter it is therefore impossible to convert NOx into N2 + CO2. In order to still remove NOx from the exhaust gases, a special NOx (storage) catalytic converter with a special barium component is required. In addition to the barium component, precious metals such as platinum and rhodium are also present in this catalytic converter.
The three-way catalytic converter converts the CO and HC values into CO2 and H2O as described earlier. The NOx is converted by the NOx catalytic converter. Additional temperature sensors and a NOx sensor are required to continuously monitor the values.
The exhaust system shown in the image below is one that is used by, among others, VW and BMW (and by more and more other brands).
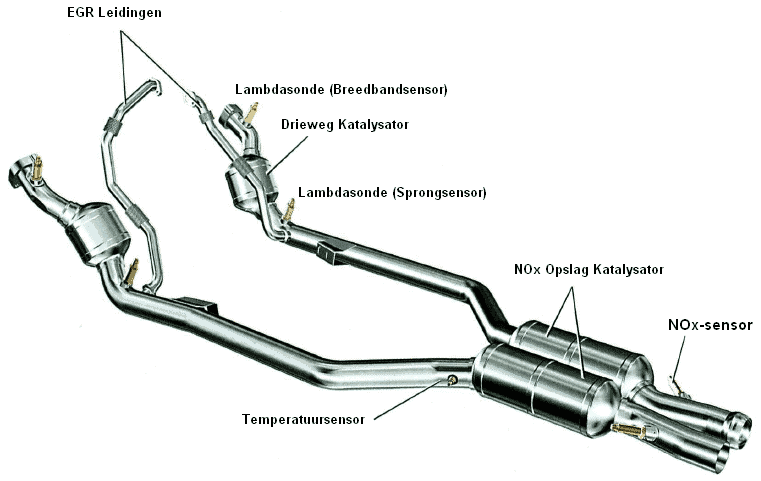
The NOx gases are stored in this catalytic converter when cold. The remaining exhaust gases can continue their way through the exhaust. During the oxygen-rich period, the NOx gases are stored in the barium component. The NOx accumulates (just like soot is stored in a particulate filter). In the long run, the catalytic converter becomes saturated. That is the moment when it is full of NOx. The catalytic converter must then be regenerated. The NOx sensor recognizes this and sends a signal to the ECU. At this point the mixture is enriched specifically to regenerate the NOx catalytic converter. This only happens when the NOx catalytic converter has reached a temperature of 800 degrees (this is recorded by the temperature sensor and also sent to the engine control unit). With the temporary enrichment, extra CO is released. With the help of this CO, the platinum and rhodium components can bring about a conversion into N2 + CO2. After regeneration, the engine will once again run on a lean mixture until the catalytic converter is saturated again.
Faults can also occur in this system. When only short trips are made with the car (which is bad for the entire car anyway), the NOx catalytic converter will not be able to reach its operating temperature either. As soon as it is saturated (full), it will need to be regenerated. Only if the temperature sensor continues to measure a temperature that is too low will the ECU never enrich the mixture. If the catalytic converter is not at operating temperature, the platinum and rhodium components cannot yet provide a conversion. At this point the engine warning light will come on and the cause will be revealed when the car is read out. The catalytic converter will then still be regenerated with the help of the diagnostic tester or a vigorous test drive. The best thing is therefore to occasionally drive a long distance (for example 50 km or more on the motorway) and preferably at an increased engine speed. The catalytic converter will then easily be able to reach its operating temperature.
Nowadays, the SCR (Selective Catalytic Reduction) catalytic converter is used in diesel engines. In this SCR catalytic converter, NOx is also stored, but an AdBlue dosing system has also been added.
Aging and causes:
- Petrol: A three-way catalytic converter can only operate with unleaded petrol. If leaded petrol is used after all, it adheres in a thin layer to the precious metal, as a result of which contact with the exhaust gases decreases and after a while is no longer even possible. No chemical reaction can then take place anymore. The catalytic converter is now inoperative and must be replaced. That is an expensive matter. Leaded petrol was added to achieve a certain knock limit (ping limit). Because knock sensors are used nowadays, lead has been removed from the fuel.
- Oil also has a devastating effect on the internals. With severe oil leakage along, for example, the piston rings, valve guides or the turbo, a lot of oil can end up in the catalytic converter. Due to the oil, a layer also adheres to the precious metal, which then loses its effect.
- Short trips: If many short distances are driven, the catalytic converter rarely if ever reaches its operating temperature. The unburned HC (petrol) residues attach themselves to the ceramic surface. If the car is driven for a long stretch, these HC residues are still burned off. If only short stretches continue to be driven, these HC residues will also adhere to the internals, causing the catalytic converter to lose its effectiveness over time.
The second lambda sensor (the switching sensor) often measures whether the catalytic converter has properly converted the gases. If the catalytic converter ages or if the internals are defective, this second lambda sensor will measure it. A warning light will then come on on the dashboard. Replacing the catalytic converter is then necessary. More information about the lambda sensor can be found on the Lambda sensor page.
