Introduction:
The task of the battery is to supply power to the consumers at times when the alternator supplies no or insufficient energy, for example when starting the engine. The battery is a buffer in which energy is stored. The energy supplied by the alternator is stored in the battery and must be released again when needed. Because electrical energy is difficult to store, the electrical energy supplied by the alternator is converted into chemical energy. When the battery then has to supply electrical energy to the consumers, the chemical energy is converted back into electrical energy.
If the car battery is in good condition but is still empty again after standing still for a few hours, there may be a clandestine consumer.
Operation:
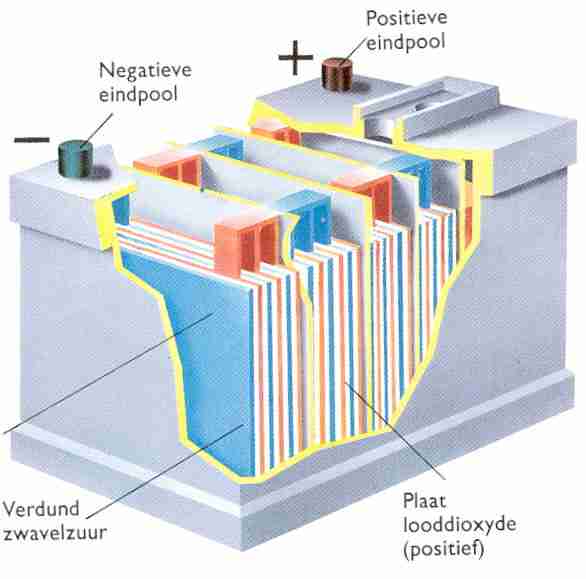
Inside the battery there are several thin lead plates in a container with sulfuric acid. The lead forms a bond with sulfur. A chemical reaction then takes place. The lead is converted into lead sulfate (PbSO4).
Diluted sulfuric acid is a mixture of sulfuric acid and demineralized (purified) water. Diluted sulfuric acid is often called electrolyte. When the lead plates are connected to a charging device the lead plates will undergo a change. The plate that is connected to the negative terminal releases sulfur to the electrolyte. The lead sulfate is converted into porous lead. The plate that is connected to the positive terminal absorbs oxygen from the electrolyte and releases sulfur to the electrolyte. After charging, this plate contains lead dioxide (PbO2). Due to the above-mentioned process, a potential difference arises between the positive and negative plate.
When a consumer is connected to the lead plates that have been charged in the aforementioned way, a current will start to flow. In this process, the lead dioxide of the positive plate is converted back into lead sulfate. The porous lead of the negative plate is also converted into lead sulfate. So during charging and discharging of the batteries there is a change in the positive and negative plates (chemical action). The electrolyte also undergoes a change during charging and discharging. When the battery is discharged, the positive and negative plates consist of lead sulfate. The sulfur used to form lead sulfate has been extracted from the electrolyte. The electrolyte of a discharged battery therefore has a low sulfur content. In a charged battery, the lead sulfate from the plates has been released back into the electrolyte. The electrolyte then has a high sulfur content. Because the sulfur particles are the heavier particles in the electrolyte, the specific gravity of the electrolyte increases as the state of charge of the battery increases. In a fully charged battery the electrolyte has a specific gravity of 1280 kg/m3. When the battery is fully discharged, the electrolyte has a specific gravity of 1140 kg/m3. For comparison: water has a specific gravity of 1000 kg/m3.
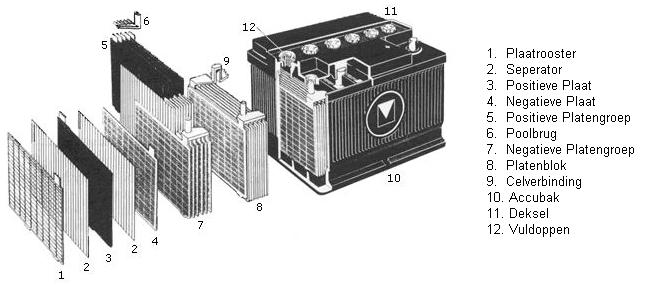
Construction of the battery:
Batteries are made up of a number of cells, each cell containing a number of positive and negative plates. Each cell has a voltage of approximately 2 V. A 12 V battery has 6 cells connected in series. The positive and negative plates are separated by separators.
Positive and negative plates:
The positive plates are connected to the positive terminal, the negative plates to the negative terminal. To prevent connection errors, both terminals are marked, and the positive terminal always has a larger diameter than the negative terminal. The positive and negative plates are interconnected by means of a bridge piece. The plates consist of a grid of lead alloy. The grids are filled with paste (a mixture of lead powder, sulfuric acid and various additives). The separators are made of plastic and cellulose. During the energy conversion in the battery, more heat is generated at the positive plate than at the negative plate. To prevent the positive plate from warping, the positive plate is always placed between two negative plates.
Battery cells:
All cells of the battery are filled with the so-called electrolyte, a mixture of distilled water and sulfuric acid. Distilled (also called demineralized) water is water from which contaminants such as lime and chlorine compounds have been removed. On older batteries, the cells are equipped with filler openings. Through these openings, demineralized water can be topped up. The filler opening can be closed with a filler cap. On newer batteries, filling is no longer possible. These are maintenance-free batteries where the water consumption is so low that topping up is not necessary.
Charging / discharging:
The state of charge of a battery can be measured with a hydrometer. A good battery charger automatically reduces the current when the charging voltage becomes higher than 2.35 V per cell (i.e. approx. 14 V with a 12 V battery). If this value is exceeded, the water molecules are split into oxygen and hydrogen, which creates hydrogen gas. If a lot of this gas is produced, it forms an explosive mixture (detonating gas).
- Normal charging:
With normal charging, the capacity of the battery is restored to 100%. The size of the charging current is 5 to 10% of the capacity. A battery with a capacity of 40 Ah is charged with a charging current of 2 to 4 A during normal charging. - Fast charging: Batteries that have become completely discharged quickly can be partially recharged by means of fast charging. The charging current is 30 to 50% of the capacity of the battery. With a battery with a capacity of 40 Ah, the charging current is 12 to 20 A. Fast charging is not used very often. Many fast chargers can also be used as a starting aid and normal charger.
- Trickle charging: When a battery is not used for a longer period of time, voltage loss occurs due to self-discharge. By constantly connecting a trickle charger to the battery, the battery is always kept fully charged. The charging current is approximately 0.1% of the capacity of the battery. A battery with a capacity of 40 Ah is then charged with a current of 0.04 A. There are battery chargers that automatically switch to trickle charge at the end of normal charging.
- Buffer charging: With buffer charging, the consumers and the charging device are both connected to the battery. The charger supplies such a current that the battery remains practically full. The battery supplies the peak current to the consumers. Buffer charging takes place when the alternator charges the battery and at the same time supplies current to the consumers. The alternator has a voltage regulator that is set to 14.4 V for a 12 Volt system. After starting, the alternator charges quickly for a while. While driving, the charging current decreases sharply. When the battery is fully charged, the charging current becomes so small that the charger only keeps the battery charged.
When the car is in a garage, it is good to keep the battery on the trickle charger. The battery then has a longer service life than a battery that is often discharged heavily for long periods, and is then quickly recharged by the alternator. A battery becomes discharged if a consumer remains switched on with the engine switched off (such as the lights). When a battery is deeply discharged (the battery is completely empty), the battery becomes internally damaged. Its service life is drastically shortened.
Capacity:
The capacity of the battery is the amount of electrical energy that the battery can contain at most. The capacity is expressed in Ah (ampere-hour). The capacity is determined on the basis of the test results. Example: A battery has a capacity of 60 Ah. This battery can supply a current of 3 A for 20 hours. (60 Ah : 20 h = 3 A). The terminal voltage will not drop below 1.75 V per cell during this.
Cold cranking current:
In general it can be assumed that the value of the cold cranking current is 4 to 5 times the capacity of the battery. The cold cranking current provides information about the speed at which the battery can supply electrical energy. For starter batteries used in cars, the cold cranking current is even more important than the capacity. The cold cranking current decreases sharply as the temperature drops. This is because the chemical reactions proceed much more slowly at a lower temperature. The conditions under which the cold cranking current is measured are predetermined.
According to DIN standards: the cold cranking current is the maximum current that the battery can supply at a temperature of 255 K (-18 degrees) for a certain period of time, at a sufficient voltage:
- After 30 sec. of discharging with the cold cranking current, the terminal voltage must still be at least 1.5 V per cell.
- After 150 sec. of discharging with the cold cranking current, the terminal voltage must still be at least 1 V per cell.
Disconnecting the battery terminals:
For certain jobs (think of the airbags, starter motor, alternator) the battery must be disconnected. Otherwise a short circuit may occur, or an airbag may be unintentionally activated. In these cases it is sufficient to remove the negative terminal. The positive terminal may remain on the battery. Never remove only the positive terminal! If it touches the bodywork (which serves as ground and is therefore connected to the negative terminal), a short circuit will occur. When the battery is removed, the negative terminal must always be removed first, and only then the positive terminal.
A battery must never be disconnected with the engine running. Modern engines are fully electronically controlled. The electronics can be severely damaged by the peak currents coming from the alternator.
In the past, a (non-electronically controlled) diesel engine could be disconnected in this way, because the fuel pump was mechanically driven and the injectors opened at a certain injection pressure. Due to the mechanical operation, the engine could continue running without a battery after starting.
Starting with jump leads:
If the battery is empty, it must be charged in order to be able to start the engine again. This can be done by connecting the battery to another car using jump leads. It is important to use good (thick) jump leads. Thin cables create a lot of resistance at high currents and therefore become very hot. There is a risk that a heavier / larger engine cannot be started with cables that are too light.
The order of connection is important; never connect the positive (red) and negative (black) cables to one battery at the same time, because this can quickly cause a short circuit when the contacts on the other side of the cable touch each other. Therefore, follow this order:
- Connect the negative cable to one car and the other end of the negative cable to the other car.
- Only then connect the positive cable to one car, and then to the other. It does not matter whether the positive and then the negative cable is connected first, or vice versa.

Both batteries are now in parallel with each other. When the batteries are connected in parallel, the voltage simply remains 12 V. So it is not the case that the total battery voltage is now 24 volts. That would be the case if the batteries were connected in series, which is what happens in, for example, electric / hybrid vehicles. For more information about series and parallel circuits (using resistors as an example), see the page current, voltage, resistance.
Now that the battery cables are connected, the alternator of the ‘charging’ car charges the empty battery. It is best to leave it like this for a minute, because otherwise it is possible that the engine still cannot be started, especially if it is a heavy diesel engine. After a minute (or longer) the car with the empty battery can be started.
The procedure when disconnecting the jump leads is also important; because the car that provides the starting aid to the other car is still transferring a lot of charging current to the empty battery via the jump leads, it is not good to remove the jump leads all at once. The charging current / voltage is very high during charging, but when disconnecting a cable, the current has nowhere to go except into the electronics of the car itself. A current spike then occurs, which can also affect the control units. This problem can be prevented by switching on all heavy consumers in the charging car (the car that is charging the empty battery). Think of the rear window defroster, lights, optional seat heating, etc. When disconnecting a jump lead, the peak current can be distributed among these components which already draw a lot of current. The control units are then spared. Disconnecting the jump leads is done in the same order as connecting them; first the positive or negative cable of both cars, and only then the other. Never remove both from one battery at the same time.
The best way to charge an empty battery is with a battery charger, because an alternator charges it with the maximum charging current. A battery charger adjusts the charging current to the condition of the battery. When a battery is deeply discharged (so when the battery voltage has dropped below 6 Volts) it becomes internally damaged. Its service life is drastically shortened.
Related page:
